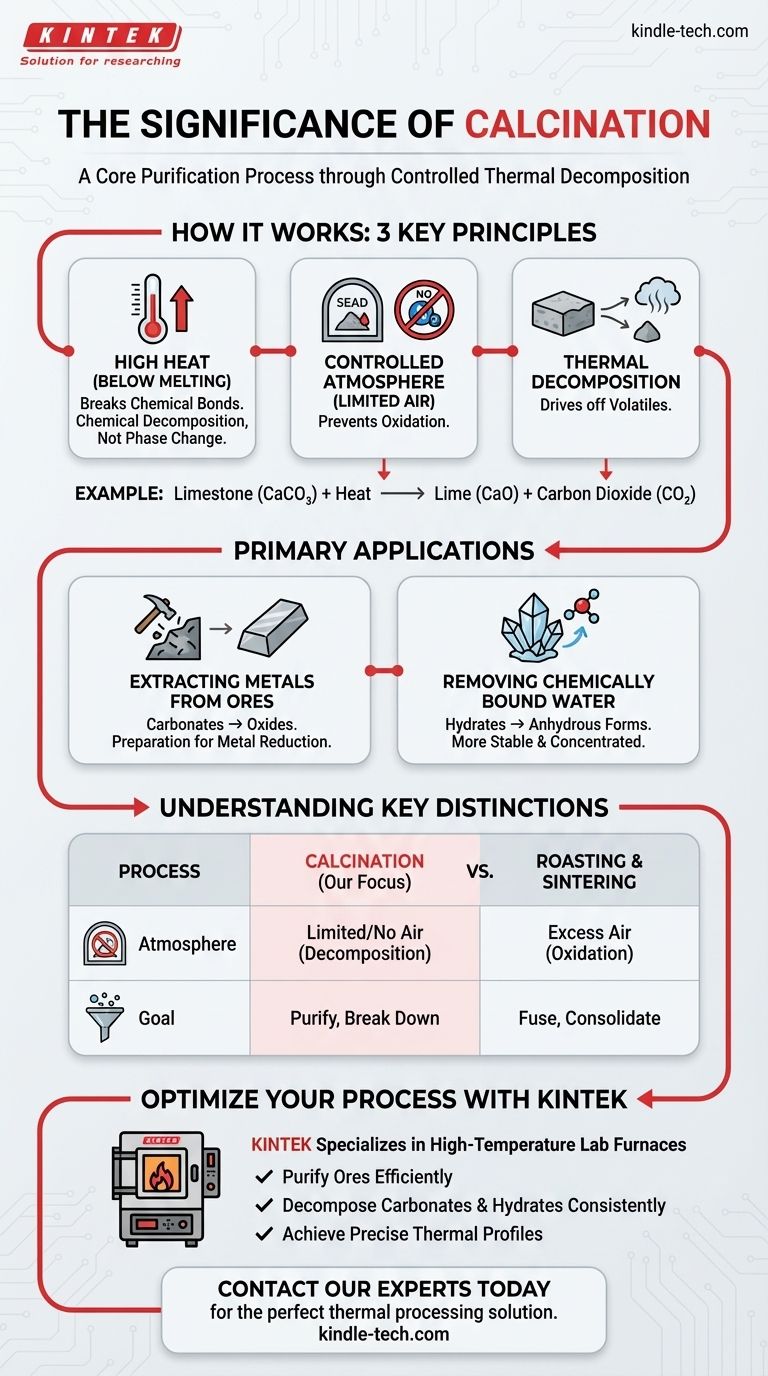
Im Kern ist die Kalzinierung ein Reinigungsprozess. Sie nutzt hohe Hitze – sorgfältig kontrolliert, um unter dem Schmelzpunkt des Materials zu bleiben –, um chemische Veränderungen hervorzurufen. In einer Umgebung mit wenig bis keiner Luft treibt dieser Prozess flüchtige Substanzen wie Kohlendioxid oder Wasser aus und verwandelt ein Rohmaterial in eine konzentriertere und stabilere Form.
Die wahre Bedeutung der Kalzinierung liegt nicht nur im Erhitzen, sondern in der kontrollierten chemischen Umwandlung. Sie dient als kritischer vorbereitender Schritt, um Verbindungen abzubauen und Verunreinigungen zu entfernen, wodurch ein Rohmaterial für nachfolgende Prozesse wie die Metallgewinnung geeignet wird.

Wie die Kalzinierung grundsätzlich funktioniert
Die Kalzinierung ist eine präzise Wärmebehandlung, die darauf abzielt, die chemische Zusammensetzung eines Materials zu ändern, nicht seinen physikalischen Zustand von fest zu flüssig. Dies wird durch drei Schlüsselprinzipien erreicht, die zusammenwirken.
Die Rolle hoher Hitze (unterhalb des Schmelzpunkts)
Der Prozess beruht auf der Zufuhr ausreichender thermischer Energie, um spezifische chemische Bindungen innerhalb einer Verbindung aufzubrechen.
Indem die Temperatur unterhalb des Schmelzpunkts gehalten wird, bleibt das Material fest. Dies stellt sicher, dass das Ziel die chemische Zersetzung und keine Phasenänderung ist.
Die kontrollierte Atmosphäre (begrenzte Luft)
Die Kalzinierung wird typischerweise in Abwesenheit oder bei sehr begrenzter Luftzufuhr durchgeführt.
Dies ist entscheidend, da es Oxidation oder Verbrennung verhindert. Das Ziel ist es, das vorhandene Material abzubauen, nicht es mit Sauerstoff in der Luft reagieren zu lassen.
Das Ergebnis: Thermische Zersetzung
Die Hitze liefert die Energie, die benötigt wird, um das Material in zwei Teile zu zersetzen: den gewünschten Feststoff und ein flüchtiges Gas, das ausgetrieben wird.
Ein klassisches Beispiel ist die Kalzinierung von Kalkstein (Calciumcarbonat, CaCO₃), der sich in Branntkalk (Calciumoxid, CaO) und Kohlendioxid (CO₂) Gas zersetzt. Der feste Branntkalk ist das wertvolle Produkt.
Die Hauptanwendungen der Kalzinierung
Dieser Prozess ist ein Eckpfeiler der Materialverarbeitung, insbesondere in der Metallurgie und der chemischen Fertigung. Sein Hauptzweck ist die Reinigung und Vorbereitung.
Gewinnung von Metallen aus Erzen
Die Kalzinierung ist ein entscheidender erster Schritt zur Verarbeitung vieler Mineralerze, insbesondere von Carbonaten und Hydraten.
Das Erhitzen eines Metallcarbonaterzes entfernt das Kohlendioxid und hinterlässt ein konzentrierteres Metalloxid. Dieses Metalloxid lässt sich dann in einem späteren Schritt viel leichter zu dem endgültigen, reinen Metall reduzieren.
Entfernung von chemisch gebundenem Wasser
Viele Mineralien existieren als Hydrate, was bedeutet, dass Wassermoleküle in ihre Kristallstruktur eingebunden sind.
Die Kalzinierung liefert die Energie, um dieses gebundene Wasser auszutreiben und das Material in seine wasserfreie (anhydrische) Form umzuwandeln, die oft stabiler und konzentrierter ist.
Wichtige Unterscheidungen verstehen
Um die Bedeutung der Kalzinierung vollständig zu erfassen, ist es wichtig, sie von anderen gängigen thermischen Prozessen zu unterscheiden. Eine Verwechslung dieser Prozesse kann zu falschen Ergebnissen führen.
Kalzinierung vs. Rösten
Der Hauptunterschied ist die Anwesenheit von Luft. Kalzinierung erfolgt mit wenig bis keiner Luft, um eine Zersetzung zu bewirken.
Rösten hingegen wird mit einem Überschuss an Luft durchgeführt, um gezielt eine Oxidationsreaktion hervorzurufen, wie die Umwandlung eines Metallsulfids in ein Metalloxid.
Kalzinierung vs. Sintern
Diese Prozesse haben entgegengesetzte Ziele. Kalzinierung zerlegt Verbindungen und treibt Verunreinigungen aus.
Sintern nutzt Hitze, um kleine Partikel ohne Schmelzen zu einem einzigen, festen Stück zu verschmelzen. Es ist ein Prozess der Konsolidierung, nicht der Reinigung.
Die richtige Wahl für Ihr Ziel treffen
Die Anwendung des korrekten thermischen Prozesses ist entscheidend, um die gewünschten Materialeigenschaften zu erzielen. Ihr Ziel bestimmt, ob die Kalzinierung der richtige Ansatz ist.
- Wenn Ihr Hauptaugenmerk auf der Reinigung eines Erzes liegt: Die Kalzinierung ist die ideale Methode zur Entfernung flüchtiger Bestandteile wie CO₂ oder H₂O aus Mineralkarbonaten und Hydraten.
- Wenn Ihr Hauptaugenmerk auf der chemischen Umwandlung liegt: Verwenden Sie die Kalzinierung, um ein Rohmaterial thermisch in ein reaktiveres oder nützlicheres Zwischenprodukt zu zersetzen, z. B. die Umwandlung von Kalkstein in Branntkalk für die Zementproduktion.
- Wenn Ihr Ziel das Verschmelzen von Partikeln oder die Reaktion eines Materials mit Luft ist: Sie benötigen einen völlig anderen Prozess, wie z. B. Sintern zum Verschmelzen oder Rösten zur Oxidation.
Letztendlich ist die Kalzinierung ein grundlegendes Werkzeug zur Kontrolle der chemischen Reinheit und Reaktivität anorganischer Materialien.
Zusammenfassungstabelle:
| Schlüsselaspekt | Beschreibung |
|---|---|
| Primäres Ziel | Chemische Zersetzung und Reinigung, nicht Schmelzen. |
| Atmosphäre | Begrenzte oder keine Luft, um Oxidation zu verhindern. |
| Wichtige Eingaben | Rohmaterialien wie Metallcarbonate oder Hydrate. |
| Wichtige Ausgaben | Gereinigte Feststoffe (z.B. Metalloxide) und ausgetriebene flüchtige Gase. |
| Hauptanwendungen | Metallgewinnung aus Erzen, Zementproduktion, Entfernung von chemisch gebundenem Wasser. |
Bereit, Ihren Materialreinigungsprozess zu optimieren?
Die Kalzinierung ist ein entscheidender Schritt zur Erzielung hochreiner Materialien in der Metallurgie und chemischen Fertigung. Die richtige Ausrüstung ist unerlässlich für eine präzise Temperaturkontrolle und eine kontrollierte Atmosphäre, um eine erfolgreiche Zersetzung ohne Oxidation zu gewährleisten.
KINTEK ist spezialisiert auf Hochtemperatur-Laboröfen, die ideal für Kalzinierungsprozesse sind. Unsere robusten und zuverlässigen Geräte helfen Ihnen dabei:
- Erze effizient zu reinigen für die Metallgewinnung.
- Carbonate und Hydrate mit konsistenten Ergebnissen zu zersetzen.
- Präzise thermische Profile für eine kontrollierte chemische Umwandlung zu erreichen.
Lassen Sie uns Ihre spezifische Anwendung besprechen. Kontaktieren Sie noch heute unsere Experten, um die perfekte thermische Verarbeitungslösung für Ihre Laboranforderungen zu finden.
Visuelle Anleitung
Ähnliche Produkte
- Elektrische Drehrohrofen-Pyrolyseofenanlage Kalzinator Kleiner Drehrohrofen Rotierender Ofen
- Elektrischer Drehrohrofen Kontinuierlicher Betrieb Kleine Drehrohrofen Heizpyrolyseanlage
- Vertikaler Hochtemperatur-Graphit-Vakuum-Graphitierungs-Ofen
- Graphit-Vakuumofen Hochwärmeleitfähige Folien-Graphitierungsöfen
- Graphitierungs-Vakuumofen für ultrahohe Temperaturen
Andere fragen auch
- Wie hoch ist der Wirkungsgrad eines Drehrohrofens? Erreichen Sie einen thermischen Wirkungsgrad von über 95 %
- Was sind die Merkmale der Gleit-, Einsink- und Rollbewegungsmodi von Schüttgütern? Optimieren Sie Ihren Drehprozess
- Was sind die Vorteile von Drehrohrofen? Erreichen Sie überlegene Gleichmäßigkeit & Effizienz
- Ist ein Drehrohrofen ein Ofen? Entdecken Sie die Hauptunterschiede für die industrielle Verarbeitung
- Bei welcher Temperatur findet die Pyrolyse statt? Ein Leitfaden zur Steuerung Ihres Produktausstoßes



















